Opsin evolution: trichromatic ancestral mammal: Difference between revisions
Tomemerald (talk | contribs) (This article presents new results on tetrachromatic and trichromativ vision during mammalian evolution using new genome projects) |
Tomemerald (talk | contribs) No edit summary |
||
| Line 17: | Line 17: | ||
Thus the 'first mammal' was certainly trichromatic and even initially tetrachromatic, depending on how it is defined along the stem relative to RHO2 loss by fossils or molecular reconstruction. This can never be settled because no datable pseudogene remnants of RHO2 remain (even with sensitive searching of the syntenic sweet spot in reconstructed ancestral genomes). The only hope here is that echidna (the only other surviving platypus relative) retains RHO2 or at least some datable fragment of it. | Thus the 'first mammal' was certainly trichromatic and even initially tetrachromatic, depending on how it is defined along the stem relative to RHO2 loss by fossils or molecular reconstruction. This can never be settled because no datable pseudogene remnants of RHO2 remain (even with sensitive searching of the syntenic sweet spot in reconstructed ancestral genomes). The only hope here is that echidna (the only other surviving platypus relative) retains RHO2 or at least some datable fragment of it. | ||
[[Image: | [[Image:opsin_platy.png|center|]] | ||
This tree was drawn by the online tool [http://iubio.bio.indiana.edu/treeapp/treeprint-sample1.html Phylodendron] using the following tree in | |||
<pre>(((((RHO1..x....x...SWS1.LWSab..human,RHO1..x....x...SWS1.LWS....elephant), | |||
This tree was drawn by the online tool [http://iubio.bio.indiana.edu/treeapp/treeprint-sample1.html Phylodendron] using the following tree in Newick format: | |||
<pre> | |||
(((((RHO1..x....x...SWS1.LWSab..human,RHO1..x....x...SWS1.LWS....elephant), | |||
RHO1..x....x...SWS1.LWS....opossum),RHO1..x...SWS2.frag.LWS....platypus)[RHO1..x...SWS2.SWS1.LWS], | RHO1..x....x...SWS1.LWS....opossum),RHO1..x...SWS2.frag.LWS....platypus)[RHO1..x...SWS2.SWS1.LWS], | ||
(RHO1.RHO2.SWS2.SWS1.LWS....chicken,RHO1.RHO2.SWS2.SWS1.LWS....lizard)) | (RHO1.RHO2.SWS2.SWS1.LWS....chicken,RHO1.RHO2.SWS2.SWS1.LWS....lizard)) | ||
[stem],RHO1.RHO2.SWS2.SWS1.LWS....frog);</pre> | [stem],RHO1.RHO2.SWS2.SWS1.LWS....frog); | ||
</pre> | |||
| Line 146: | Line 150: | ||
0 FRNCIMQLFGKKVDDGSELSSTSRTEVSSVSSVSPA* 0 | 0 FRNCIMQLFGKKVDDGSELSSTSRTEVSSVSSVSPA* 0 | ||
</pre> | </pre> | ||
[[Category: | [[Category:Comparative Genomics]] | ||
Revision as of 19:15, 20 November 2007
Proof that ancestral mammal had trichromatic vision
(open source article originated 7 Sept 07 by TH Pringle)
Textbooks sometimes portray evolution as unidirectional progress chugging along steadily to a pinnacle of human innovative perfection, even as more primitive creatures drop by the wayside for lack of forward-looking vision of manifest destiny. However the newly available comparative genomics of opsins -- the photon receptors underlyig vision -- clearly refute those notions. Evolution only lives for the moment -- opsin genes are retained on a "use or lose it" basis. A fully subterranean mammal such as the blind mole rat has no selective pressure on its opsin genes to weed out deleterious mutations. Today those are all rapidly deteriorating pseudogenes.
Gordon Walls proposed in 1942 that mammals -- but not birds and lizards -- experienced a prolonged bottleneck of strictly nocturnal lifestyle during which certain vision genes were not used and so lost irrevocably to mutation. Upon returning to sunlight, mammals had to go forward with distinctly inferior two color vision. Some marsupials and placentals (including human) have regained trichromatic vision by various mechanisms. However despite 125 million years of playing catchup, no placental mammal has regained the tetrachromatic color vision or colored oil droplet spectral enhancers enjoyed by the amniote common ancestor.
Thus color vision in placental mammals is inferior in every way not only to living fish, turtles, frogs, lizards, and birds but also inferior to a shared common ancestor from 450-150 million years ago! In other words, 300 million years of evolutionary "progress" was lost in "advanced" theran mammals. It's possible to have much better color vision than human and most vertebrate species -- extant and extinct -- do.
The advent of the genomic era has vastly improved our prospects for working out the long evolutionary history of photoreception -- ciliary opsin genes can be extracted from the 57 deuterostome genomes available (as of November 2007) and visual capabilities of last common ancestors rather safely deduced. The newly sequenced platypus genome is especially informative to the history of color vision
The platypus genome project establishes it does not encode any ancient, ancestral, antiquated, archaic, dead-end, failed experiments, fossil, frozen, primitive, primordial, relic, retro, or stationary genes. Indeed by any objective measures of evolutionary distance from the common ancestor with humans (such as lower percent identity of the average selected coding gene), the platypus is more highly evolved. Furthermore, the platypus retains hundreds of genes (shared in conserved syntentic position with bird and lizard) that are lost in the human. That's not surprising because the platypus is a modern organism highly successful in its current preferred habitat but rather because living fossils, strictly defined, cannot exist since no species is immune to mutational processes (followed by drift and selection that fix them).These genes represent functionality conserved in ancestral tetrapods for hundreds of millions of years that subsequently degenerated in placentals.
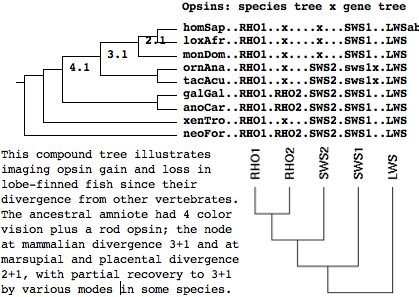
Reasoning directly from the platypus genome, the ancestral mammal was tetrachromatic (4 cone opsins plus rod opsin) -- indeed all those genes had originated not later than the Ordovician (in the common stem ancestor with jawless fish). The diagram below shows the relevent portion of the phylogenetic tree; the horizontal branch labelled 'stem' links the bird + lizard divergence node to platypus divergence. At the platyus node, the common ancestor had RHO1, no RHO2, SWS2, SWS1, and LWS. This implies RHO2 was lost at some point on the stem. Prior to that loss, the ancestor had a full complement of imaging opsins (ie RHO1, RHO2, SWS2, SWS1, and LWS).
Thus the 'first mammal' was certainly trichromatic and even initially tetrachromatic, depending on how it is defined along the stem relative to RHO2 loss by fossils or molecular reconstruction. This can never be settled because no datable pseudogene remnants of RHO2 remain (even with sensitive searching of the syntenic sweet spot in reconstructed ancestral genomes). The only hope here is that echidna (the only other surviving platypus relative) retains RHO2 or at least some datable fragment of it.
This tree was drawn by the online tool Phylodendron using the following tree in Newick format:
(((((RHO1..x....x...SWS1.LWSab..human,RHO1..x....x...SWS1.LWS....elephant), RHO1..x....x...SWS1.LWS....opossum),RHO1..x...SWS2.frag.LWS....platypus)[RHO1..x...SWS2.SWS1.LWS], (RHO1.RHO2.SWS2.SWS1.LWS....chicken,RHO1.RHO2.SWS2.SWS1.LWS....lizard)) [stem],RHO1.RHO2.SWS2.SWS1.LWS....frog);
Opsins illustrate how the platypus genome successfully bridges a long-standing gap in our understanding of mammalian color vision evolution. However proof of the opsin evolution story requires more than just handwaving -- the argument revolves around highly technical genome annotation issues as explained shortly. For starters vertebrate genomes are incomplete at this time, so absence of evidence in an assembly does not constitute evidence of absence in the genome, but more commonly lack of coverage, even consulting unused raw traces (33,353,710 total for platypus).
Opsins don't lend themselves to simple homology-based parsimony reconstructions based on processed transcript fata because of evolutionary convergence of experimentally accessible (spectroscopic) properties and lineage-specific expansions and contractions of genes. What's needed here are complete genomes and syntenic validation of orthology, which we lack to a certain degree here in key species (eg 3 of 6 known chicken opsins are missing from its current assembly).
However using the platypus genome, it is possible to reliably reconstruct the cone opsin situation at the ancestral mammal node, building on ref 2. This is a difficult argument because platypus assembly initially exhibits only exon 5 of an opsin gene clustering to the short wavelength sensitive opsin SWS1 (as observed in ref 2). This last coding exon has the phylogenetically expected position and phase. It must be shown that the rest of platypus SWS1 has been deleted from the genome, rather than just be a coverage gap in the platypus assembly:
Close synteny analysis of the gapless platypus assembly contig 45.4 using a conserved non-coding phastCons marker and adjacent CALU gene shows that exons 1-4 do not lie in adjacent assembly gaps but were lost in a multi-kbp deletion. Exon 5 exhibits a small number of disabling non-synonymous changes despite retaining overall high (63%) sequence identity to human, consistent with millions of years of normal divergence followed by rapid neutral evolution after the deletion of exons 1-4 pseudogenized residual exon 5. The deletion can be heuristically dated to perhaps 40 mya, an estimate that echidna SWS1 sequence would greatly improve. No opsin data currently exists for echidna; they may retain functional SWS1.
It follows that ancestral monotreme resp. ancestral mammal had 3-color vision (one step down from ancestral amniote) even though the platypus itself today has 2-color vision (similar to marsupials and most placentals). (This assumes intact SWS1 was not exapted to some unrelated function; this is testable from its spectral reconstruction.)
Queries by known SWS1 genes -- but not other opsins -- return a strong match to platypus chr10:3,960,795-3,960,908, which translates to FHACIMEMMRGKLMVDDSESSSQETKTSTVSSRQVGPS* with conserved intron position and phase zero characteristic of SWS1 exon 5. The 11,205 bp contig 45.4 also contains terminal exons of long-conserved syntenic gene CALU in the expected opposite strand orientation. Total coding gene span seems short enough (3,646 bp in opossum, 3,248 bp in human, 11,959 bp in anole) that the contig might be expected to contain some or all of exons 1-4 as well, even adjusting contig length to reflect observed lineage-specific retroposons of platypus. However exons 1-4 are missing from the contig and indeed the assembly. Furthermore missing exons cannot be found among GenBank transcripts now raw trace reads which include singleton reads and reads from other centers not used in the assembly.
This raises the question whether platypus lacks full length SWS1 or whether exons 1-4 merely lie in nearby gaps of the current 6x assembly. Possibly earlier introns have expanded so much that exons 1-4 have been pushed off the contig into these gaps. However contig 45.4 contains the phylogenetically conserved non-coding phastCons element lod109 (ref 3) at its extreme 3' end chr10:3,964,439-3,964,588 (the contig ends at 3,964,658!).
The UCSC genome browser provides putative orthologs of lod109 in marsupial and placental. These blat as standalone queries into full syntenic position with matching strand orientation with respect to marsupial and mouse where they are named lod15+lod45 and lod78 respectively. Here phastCons strand convention, which is inherently arbitrary, can be taken as positive for platypus and carried forward to mouse and oppossum. CALU is positive but SWS1 exon 5 is negative in all three species.
Thus exons 1-4 of platypus are required to lie between SWS1 exon 5 and lod109, a region spanned by a single gapless contig, but they do not. This requires either a deletion or translocation elsewhere (to a trace read and assembly gap). A local inversion might move exons 1-4 into an assembly gap but that would disrupt reading frame. Implausibly, the missing exons could be translocated into an adjacent bridged gap of estimated modest size preceding the right-flanking gene NAG6 while retenting strand; this is incompatible with all four exons remaining recalcitrant to repeated attempts at genomic amplification (ref 2) and still missing in 6x coverage.
In summary, SWS1 is non-functional in platypus because the highly conserved exons 1-4 critical to opsin structure and function have been deleted. Conceivably this could be a copy-number polymorphism specific to the individual female platypus chosen for the genome project -- a seemingly viable concern given such polymorphisms are common for the tandem chrX opsin array in human. [Note the current human assembly shows 3 tandem opsins on chrX!] However platypus SWS1 does not lie in tandem array that might foster frequent inhomogeneous recombination. Human colorblindness involving loss of orthologous SWS1 is quite rare.
Further close analysis of exon 5 shows the deletion event lies between ancient and recent:
Alignment of orthologous exons from 19 species phylogenetically bracketing platypus establish an arginine anomalously replacing conserved cysteine and a one residue deletion in platypus but otherwise very respectable percent identity that is not compatible with gene loss 166 mya ago (date for divergence taken in platypus paper). Non-selected pseudogenized coding dna does not leave detectable remnants at this age -- indeed no remnants can be detected of amniote RHO2 in mammals nor SWS2 in therans even focusing the search on syntenic dna, ref 2 and here.
>SWS1_xenTro FRGCIMETVCGRPMTDDSSVSSTSQKTEVSTVSSSQVSPA* amphibian >SWS1_anoCar FRACILETVCGKPMSDESDVSSSAQKTEVSSVSSSQVSPS* lizard >SWS1_pheMad FRGCIMEMVCGKPMSDDSEASTS-QKTEVSSVSSSQVSPS* lizard >SWS1_gekGek FRGCILEMVCGKTMAEESEVSSASQKTEVSSVSSSQVGPS* lizard >SWS1_utaSta FRACIMETVCGKPMTDESDVSSSAQKTEVSSVSSSQVSPS* lizard >SWS1_galGal FRACIMETVCGKPLTDDSDASTSAQRTEVSSVSSSQVGPT* bird >SWS1_taeGut FRACIMETVCGRPMTDDSEVSSSAQRTEVSSVSSSQVGPS* bird >SWS1_phaCar FRACIMETVCGKPMADDSEASSSAQRTEVSSVSSSQVSPS* bird >SWS1_ancMam FHACIMEMVCGKPMTDDSdVSSS-QKTEVSTVSSSQVGPS* ancestral mammal >SWS1_ornAna FHACIMEMMRGKLMVDDSE-SSS-QETKTSTVSSRQVGPS* monotreme >SWS1_monDom FHACIMEMVCRKPMTDDSDVSSS-QKTEVSAVSSSQVGPT* marsupial opossum Didelphimorphia >SWS1_macEug FHACIMEMVCRKPMTDDSEASSS-QKTEVSTVSSSQVGPS* marsupial wallaby Diprotodontia >SWS1_smiCra FHACIMEMICKKPMTDDSETTSS-QKTEVSTVSSSQVGPS* marsupial dunnart Dasyuromorphia >SWS1_setBra FHACIMEMVCRKPMTDDSEASSS-QKTEVSTVSSSQVGPS* marsupial quokka Diprotodontia >SWS1_isoObe FHACIMEMICRKPMTDDSETSSS-QKTEVSTVSSSQVSPS* marsupial bandicoots Peramelemorphia >SWS1_canFam FRACIMEMVCGKSMTEDSEMSSS-QKTEVSTVSPSQVGPN* placental >SWS1_susScr FRACIMEMVCGKPMTDESDMSSS-QKTEVSTVSSTQVGPN* placental >SWS1_homSap FQACIMKMVCGKAMTDESDTCSS-QKTEVSTVSSTQVGPN* placental >SWS1_sciBol FRACIMEMVCGKAMTDESDISSS-QKTEVSTVSSSQVGPN* placental >SWS1_cavPor FRACIMELVCRKPMADESDMSTS-QKTEVSAVSSSKVGPN* placental >SWS1_musMus FRACILEMVCRKPMADESDVSGS-QKTEVSTVSSSKVGPH* placental Consensus FraCImEmvcgkpMtD#S#.sss.QkTevStVSSsqVgP.*
The anomalous arginine can be validated in raw trace data, necessary because the trace assembly process resolves discrepancies by quality scores weighting that might disadvantage a read more accurate locally at the cysteine position. Viewing original data requires blastn against separate platypus WGS and OTHER databases at the NCBI trace archive repository. Some 752,000 additional traces from TIGR and NISC are found there in addition to 3,595,283 cDNA WUGSC traces. These numbers are a significant addition to the 27,607,516 conventional WGS traces used (but not used up) in the current platypus assembly.
The usual error types occur in these traces but none would result in R --> C and similarly for the 3bp indel. This cysteine is deeply conserved in homologous position in other opsins such as RHO1, SWS2, and LWS though there is 6 residues of spacing in the latter two families instead of 5 here.Therefore arginine, manifestly unsuited for a disulfide bond, is not acceptable structurally in this position.
The amino acid in platypus is a bit unusual at four other sites, highlighted in red in the alignment. Some of these changes may be incapacitating but others tolerated or even advantageous. While other species have their anomalies as well, platypus is a real outlier in this regard which can be seen for example by MultiAlign tree construction.
The alignment contains a heuristic prediction of exon 5 in the last common ancestor of mammals that is useful in counting changes in descendant lineages. Overall platypus exon 5 has evolved anomalously in that its variations are not concentrated at commonly variable positions but disproportionately at conserved ones, a pattern characteristic of a pseudogene. Opsin change is difficult to assess because to be retained over long timeframes, SWS1 and SWS2 in the same species may push each other apart (subfunctionalize) at significant sites that affect absorption spectra (eg color vision) rather than just drift at soft positions.
Platypus exon 5 can only have become nonfunctional fairly recently -- retaining 63% identity to human given 332 mya of round-trip evolutionary time is quite respectable and implies long periods of conserved functionality. (Chicken/human have 65% identity.) This degree of conservation is incompatible with ancestral monotreme already having pseudogenized SWS1 from consideration of neutral rates. Thus it appears that a platypus ancestor, possibly prior to echidna divergence at 25 mya (ref 4), still had functional SWS1 and so trichromatic cone vision. That would also be true further back at the ancestral mammalian node because platypus retains SWS2 and LWS, still in amniote-syntenic tandem position at Ultra401:228519-263967 (blat genome or see ref 2).
In summary, the preferred scenario is platypus ancestors possessing a functional SWS1 gene evolving at a moderate rate until the fairly recent deletion of exons 1-4. After the deletion the remaining exon 5 had no constraints and so evolved at the faster neutral rate of pseudogenic dna.
How recent was this deletion? That would require partitioning changes in platypus exon 5 since divergence from ancestral mammal between drift and selective change during the functional era from subsequent neutral change in the pseudogene era, based on anomalous character and what is known generally about opsin evolution and neutral rates in general. That calculation would greatly benefit from determining the sequence and status of exon 5 in echidna. If echidna still had functional SWS1 and the divergence time was 25 mya, the deletion event in platypus must post-date that. On the other hand, if echidna also contained the indel, the departure of its exon 5 sequence from platypus would significantly inform event partitioning and so improve dating of the indel.
It cannot be proven that intact SWS1 functioned as a cone cell color vision receptor in ancestral platypus (because opsins can be exapted to other purposes); echidna sequence might at least allow appropriateness of its adsorption spectrum to be adduced.
ref 1. Walls, GL The Vertebrate Eye and its Adaptive Radiation Cranbrook Institute 1942
ref 2. Davies WL, Carvalho LS, Cowing JA, Beazley LD, Hunt DM, Arrese CA Visual pigments of the platypus Curr Biol. 2007 Mar 6;17(5):R161-3.
ref 3. Siepel A, Bejerano G, et al. Evolutionarily conserved elements in vertebrate, insect, worm, and yeast genomes Genome Res. 2005 Aug;15(8):1034-50.
ref 4. Pettigrew JD. Electroreception in monotremes J Exp Biol. 1999 May;202(Pt 10):1447-54.
The critical conserved non-coding markers:
>lod109_ornAna+ chr10:3964439-3964588 tgctgctgatgtggcaaagaagaccacatctgtccagctacttctatcaccggctcgttggtctaggggtatgattctcgcttagggtgcgagaggtcccgggttcaaatcccggacgagccctgtttttcccacctggttttatttggg >lod109_monDom+ chr8:184,539,020-184,560,157 from platypus alignment tgctgatgatgtggcaaaaccacatctggacaactattttcaacactggctcgttggtctaggggtatgattctcgcttagggtgcgagaggtcccgggttcaaatcccggacgagcccaattttac >lod78_musMus+ chr6:29338830-29338906 from platypus alignment tgttgatgatgtggtagag-aggatacatctggccaggagtaaccgccagtggctcgttggtctaggggtatgattctcgcttagggtgcgagaggtcccgggttcaaatcccggacgagccctgcttatc
The five imaging opsin genes in modern platypus genome:
>RHO1_ornAna Ornithorhynchus anatinus (platypus) rod rhodopsin 0 MNGTEGQDFYIPMSNKTGVVRSPFEYPQYYLAEPWQYSVLAAYMFMLIMLGFPINFLTLYVTIQHKKLRTPLNYILLNLAFANHFMVLGGFTTTLYTSLHGYFVFGPTGCNIEGFFATLG 1 2 GEIALWSLVVLAIERYIVVCKPMSNFRFGENHAIMGVAFTWIMALACALPPLVGWSR 2 1 YIPEGMQCSCGIDYYTLRPEVNNESFVIYMFVVHFTIPMTIIFFCYGRLVFTVKE 0 0 AAAQQQESATTQKAEKEVTRMVIIMVIAFLICWVPYASVAFYIFTHQGSNFGPIFMTVPAFFAKSSAIYNPVIYIMMNKQ 0 0 FRNCMLTTICCGKNPLGDDEASATASKTEQSSVSTSQVSPA* 0 >SWS2_ornAna Ornithorhynchus anatinus (platypus) cone short blue 0 MHKTHRNLQNELPEDFFIPLPLDTDNITSLSPFLVPQTHLGGSGIFMSLAAFMFLLITLGFPINLLTVICTIKYKKLRSHLNYILVNLAVSNMLVVCVGSATAFYSFAHMYFVLGPTACKIEGFAATLG 1 2 GMVSLWSLAVIAFERFLVICKPLGNLSFRGTHAIFGCAATWVFGLAASLPPLFGWSR 2 1 YIPEGLQCSCGPDWYTTNNKWNNESYVIFLFSFCFGVPLSIIIFSYGRLLLTLRA 0 0 VAKQQEQSATTQKAEREVTKMVIVMVLGFLVCWLPYASFSLWVVTNRGQVFDLRMASIPSVFSKASTIYNPIIYVFMNKQ 0 0 FRSCMLKLVFCGKSPFGDEDEISGSSQATQVSSVSSSQVSPA* 0 >RHO2_ornAna Ornithorhynchus anatinus (platypus): gene absent from genome and PCR >SWS1_ornAna Ornithorhynchus anatinus (platypus): chr10:3,935,884-3,985,883 terminal exon fragment surviving deletion event FHACIMEMMRGKLMVDDSESSSQETKTSTVSSRQVGPS* >LWS_ornAna Ornithorhynchus anatinus (platypus) cone long LWS green 0 MTPAWNSGVYAARRRFEDEEDTTRTSVFVYTNSNNTR 1 2 DPFEGPNYHIAPRWAYNVTSLWMIFVVIASVFTNGLVLVATMKFKKLRHPLNWILVNLAVADLGETLIASTISVINQIFGYFILGHPMCVLEGYTVSLC 1 2 GITGLWSLSIISWERWIVVCKPFGNVKFDAKLAMVGIVFSWVWAAVWTAPPIFGWSR 2 1 YWPHGLKTSCGPDVFSGSSDPGVQSYMIVLMSTCCILPLSIIVLCYLQVWLAIRA 0 0 VAKQQKESESTQKAEKEVSRMVVVMILAYCFCWGPYTIFACFAAANPGYAFHPLAAALPAYFAKSATIYNPIIYVFMNRQ 0 0 FRNCIMQLFGKKVDDGSELSSTSRTEVSSVSSVSPA* 0